Introduction
The focus of the module is to assist you to analyse trends and patterns in relation to the properties of pure substances and use these to predict the properties of other pure substances. This knowledge is used to determine the ways in which substances can be separated from each other and those that allow them to remain together.
Matter can be either pure substances with distinct measurable properties (eg melting and boiling points, reactivity, strength, density) or mixtures with properties that are dependent on the identity and relative amounts of the substances that make up the mixture. The analysis of these properties has led to the expansion of the periodic table of elements and the advancement of atomic theory. This understanding has allowed for the development of complex models that have been subject to extensive peer review, and has contributed to advances in many disciplines over time.
You will use knowledge obtained from the study of the periodic table to examine trends and patterns that exist between chemical elements and atoms in order to discover that fundamental particles, and their role in the structure of an atom, give all chemicals their properties.
There are four main areas of content:
Properties of Matter
Atomic Structure and Atomic Mass
Through these areas of content, you will have the opportunity to explore the following questions:
How do the properties of substances help us to classify and separate them?
Why are atoms of elements different from one another?
Are there patterns in the properties of elements?
What binds atoms together in elements and compounds?
HOMEWORK FOR EACH LESSON:
After each lesson, use a combination of these to consolidate, clarify, and identify questions for the next lesson.
1. Create Cornell notes (template below): brainstorm the key ideas you remember from the lesson. Add detail to the outline mindmap (above) if you find that more useful. This helps with memory retention.
2 . View Col Harrison videos (will be included throughout the site) to add key points you may have missed, or not understood.
3. To complete questions, to check understanding, to identify questions you may have (add these to your Cornell notes), use:
Focus textbook relevant sections, and/or
PSM Workbook (below)
4. Use the IQ Response Practice booklet (below) to develop your ability to write exam responses.
1. Cornell notes template
2. PSM Workbook
3. Response Practice Booklet
IQ1 How do the properties of substances help us to classify and separate them?
1.1a explore homogeneous mixtures and heterogeneous mixtures through practical investigations:
using separation techniques based on physical properties
1.1a-1 View PPTs: https://drive.google.com/drive/u/1/folders/1wh9NrjWiICM38b7Qeulq0A5TDODRiHUx
States of Matter
Mixtures
1.1a-2. View the padlet (below, not mine), which shows multiple physical properties that can be used for separations. Scroll across. The final box should read opacity.
1.1a-3. Homework videos:
1.1b explore homogeneous mixtures and heterogeneous mixtures through practical investigations:
calculating percentage composition by weight of component elements and/or compounds
1.1b-1 Worksheet: Percentage composition worksheet
1.1b-2 Practicals:
Gravimetric analysis of choc-chip cookies https://drive.google.com/drive/u/1/folders/1wh9NrjWiICM38b7Qeulq0A5TDODRiHUx
Gravimetric analysis of sand / salt mixture
1.1b-3 Homework Video:
IQ2 Why are the atoms of elements different from one another?
2.1 classify the elements based on their properties and position in the periodic table through their:
- physical properties
- chemical properties
Homework videos:
Classifying elements physically: https://www.youtube.com/watch?v=PrUH2XGyafI&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP&index=4
Classifying elements chemically: https://www.youtube.com/watch?v=CIzt0pUDq7A&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP&index=7
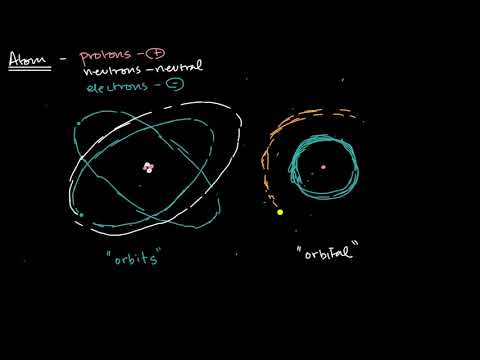
2.2 Model the atom’s discrete energy levels, including electronic configuration and spdf notation
2.2-1 Read the following (only read the text showing, continue further if you want to extend, but we will cover that in class.)
2.2-2 View PPTs:
Atomic Structure and Atomic Mass PPT
Quantum Physics PPT
2.2-3 View videos:
Atomic History: https://www.youtube.com/watch?v=rwn427EPBR0&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP&index=13
Electron configuration: https://www.youtube.com/watch?v=VWyTey2x2s8&index=10&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP
Quantum numbers Prof Dave: https://www.youtube.com/watch?v=Aoi4j8es4gQ
Orbitals explained: https://www.youtube.com/watch?v=CP35NuspGlk
spdf orbitals explained: https://www.youtube.com/watch?v=sE1IvKAijmo
Orbitals crash course: https://www.youtube.com/watch?v=cPDptc0wUYI
2.2-4 Complete activities:
Reading: http://www.chemguide.co.uk/atoms/properties/atomorbs.html
Simulation: http://employees.oneonta.edu/viningwj/sims/atomic_electron_configurations_s1.html (requires sign in)
2.2-5 Homework videos:
2.3 investigate energy levels in atoms and ions through:
- collecting primary data from a flame test using different ionic solutions of metal salts
- examining spectral evidence for the Bohr model and introducing the Schrödinger model
View PPT:
Electron Config PPT https://drive.google.com/drive/u/1/folders/1wh9NrjWiICM38b7Qeulq0A5TDODRiHUx
View videos:
Flame tests: https://www.youtube.com/watch?v=lKruq_80LKc&index=8&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP
Schrodinger model: https://www.youtube.com/watch?v=y9eb1yQ_Yw0&index=14&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP
Bohr model flame test explanation: https://www.youtube.com/watch?v=tuRnBEiQ4yA
Bohr and Schrodinger models: https://www.youtube.com/watch?v=ijFYl4zHscY
Read:
Complete activities:
2.4 calculate the relative atomic mass from isotopic composition
View PPTs: https://drive.google.com/drive/u/1/folders/1wh9NrjWiICM38b7Qeulq0A5TDODRiHUx
Atomic Structure and Mass PPT
Atomic Mass PPT
Isotopes Spectra PPT
View videos:
Isotopes: https://www.youtube.com/watch?v=kTcGkLR9dQ0&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP&index=12
Symbol notation for isotopes: https://www.youtube.com/watch?v=Gso_eNAIJdc&index=11&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP
Relative atomic mass: https://www.youtube.com/watch?v=KymWeZ6mNd8&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP&index=9
Relative atomic mass: https://www.youtube.com/watch?annotation_id=annotation_1815037825&feature=iv&src_vid=aqfQKzbTCVo&v=PrNVj8i_oDA
Complete online activities:
Average Atomic Mass Practice Problems https://www.youtube.com/watch?v=rgixSP7PxS0&index=17&list=PL0o_zxa4K1BWziAvOKdqsMFSB_MyyLAqS [10.19 mins]
2.5 investigate the basic structure of stable and unstable isotopes by examining:
- their position in the periodic table
- the distribution of electrons, protons and neutrons in the atom
- representation of the symbol, atomic number and mass number (nucleon number)
View PPTs: https://drive.google.com/drive/u/1/folders/1wh9NrjWiICM38b7Qeulq0A5TDODRiHUx
Radiation PPT
View videos:
Radiation and radioisotopes: https://www.youtube.com/watch?v=ok63ZzUQbx4&index=20&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP
View simulations:
2.6 investigate the properties of unstable isotopes using natural and human-made radioisotopes as examples, including but not limited to:
- types of radiation
- types of balanced nuclear reactions
View videos:
Applications of radioisotopes: hhttps://www.youtube.com/watch?v=HhsgpMDEKs8&index=18&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP
Nuclear reactions: https://www.youtube.com/watch?v=0FSG4-wCQYs&index=19&list=PLeFSFSJ9WqSARl70FH6vBJCVfhtu4JjKP
Review:
View PPTs: https://drive.google.com/drive/u/1/folders/1wh9NrjWiICM38b7Qeulq0A5TDODRiHUx
IQ2 Wrap up PPT